Cheerios, Gatorade, Advil, and Thousands More Recalled Over Rodent Waste Contamination, Salmonella


Nearly 2,000 products ranging from drugs, medical devices, cosmetics, dietary supplements, human food, and pet food have been pulled from shelves after the FDA found rodent droppings, rodent urine, and bird waste at a Minnesota distribution facility. Gold Star Distribution, Inc. issued a safety alert on December 26, 2025, warning that anyone who handles or consumes the affected items could become seriously ill. The recall includes brands like Cheerios, Pringles, and Advil, with products distributed across Minnesota, North Dakota, and Indiana.
Federal Inspectors Found Filth in Storage Areas

The FDA determined that Gold Star Distribution’s Minneapolis facility was operating under “insanitary conditions,” with rodent excreta, rodent urine, and bird droppings found in areas where medical devices, drugs, human food, pet food, and cosmetics were stored, federal officials said. Such conditions allow products to become contaminated when they touch dirty surfaces or are exposed to particles in the air from animal waste. The facility is located at 1000 N. Humboldt Ave in Minneapolis.
Salmonella and Leptospirosis Pose Serious Health Threats
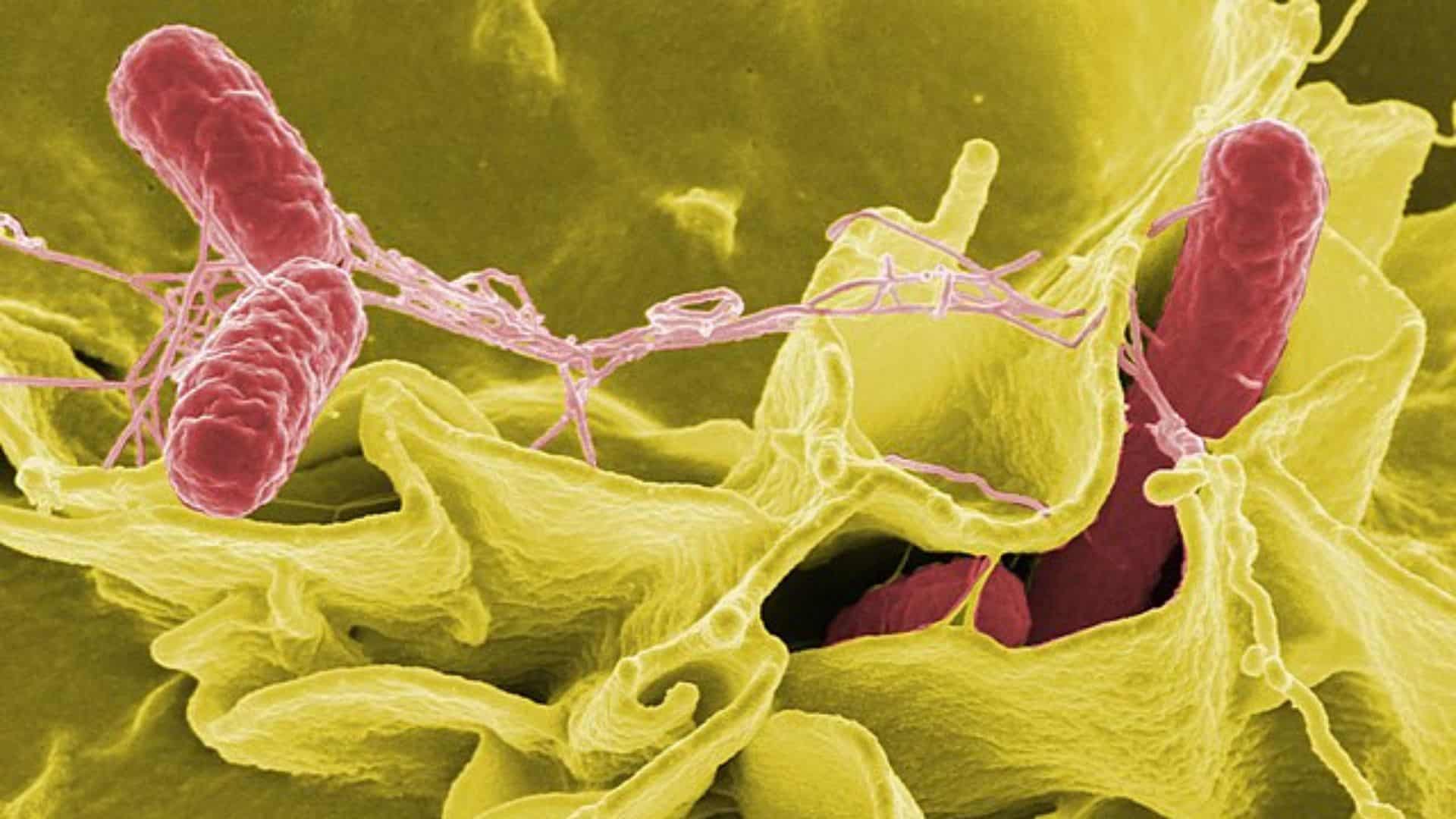
Contaminated products may harbor dangerous bacteria, including Salmonella, which can cause severe and sometimes fatal infections in vulnerable groups like infants, elderly individuals, pregnant women, and those with weakened immune systems, according to the company’s announcement. Healthy people infected with Salmonella typically experience fever, bloody diarrhea, nausea, vomiting, and abdominal pain. Rodent urine and excreta can also transmit leptospirosis through Leptospira bacteria. In rare cases, Salmonella infections can enter the bloodstream and cause infected aneurysms, endocarditis, and arthritis.
The Recall Spans Six Different Product Categories

The voluntary recall affects all FDA-regulated products held at the facility, spanning drugs, medical devices, cosmetics, dietary supplements, human food, and pet food. Contaminated medical devices could increase infection risks, while drugs and foods may cause adverse health effects if consumed. Cosmetics applied to skin or eyes might lead to irritation, infection, or other reactions, according to the company. The recall excludes frozen and refrigerated items shipped directly to retail stores by distributors or manufacturers.
Popular Brands Include Cheerios, Takis, and Coca-Cola

The comprehensive product list includes widely recognized brands like Cheerios, Pringles, Nutella, Benadryl, Aleve, Cream of Wheat, Airheads, Maruchan Ramen, Jolly Ranchers, Takis, Gatorade, Coca-Cola, and Ortega Taco Seasoning. A complete inventory appears on the FDA’s website. A spokesperson for Gold Star Distribution told TODAY.com that while no complaints or illnesses have been reported, the company is working closely with federal regulators on the voluntary action. The spokesperson said Gold Star has been in business for more than 30 years and takes this situation very seriously.
Products Were Sent to Dozens of Retail Locations

Recalled items were distributed to stores across three states, with the majority concentrated in Minnesota communities, including Minneapolis, St. Paul, Rochester, Burnsville, Bloomington, and Brooklyn Park. Products were also distributed to one store in Fargo, North Dakota, and another location in Indianapolis, Indiana. Gold Star notified all retail customers and provided instructions to destroy the products, with documentation sent back to the company. A detailed list with specific store names, addresses, and cities is available through the FDA.
Customers Must Destroy Products, Not Return Them

Anyone who purchased the affected items should destroy them immediately and verify destruction by providing a receipt to Gold Star at 1000 N. Humboldt Ave, Minneapolis, MN 55411, the company said. Customers should not ship products back to Gold Star for any reason. The company will issue refunds upon request. People experiencing health problems potentially related to the contaminated products should contact their physician or healthcare provider. Pet owners concerned about recalled pet food should contact their veterinarian.
No Illnesses Reported Yet, Company Says

No illnesses have been reported to date related to the contaminated products, according to Gold Star’s December 26 announcement. A spokesperson told TODAY.com the company takes the situation very seriously and is working closely with the FDA on the voluntary recall. The company issued the safety alert warning that anyone who handles or consumes affected products could become seriously ill.
Multiple Ways to Report Adverse Reactions

Customers can report adverse reactions or quality problems to the FDA’s MedWatch Adverse Event Reporting program online, by regular mail, or by fax. People can complete and submit reports online, download a form by calling 1-800-332-1088, or fax completed forms to 1-800-FDA-0178. The FDA’s SmartHub also helps the public navigate to appropriate forms for reporting issues with FDA-regulated products, adverse health experiences, or facility problems. For questions, customers can contact Gold Star at 612-617-9800, seven days a week, 8:00 a.m. to 5:00 p.m. Central Time.
Recall Conducted With Federal Oversight

The recall is being conducted with FDA knowledge, according to Gold Star’s announcement. The contamination stemmed from pests, including rodents, birds, and insects that left waste in storage areas. Products may have come into contact with contaminated surfaces or been exposed to harmful microorganisms. The nearly 2,000 affected products span six FDA-regulated categories and were distributed to stores across Minnesota, North Dakota, and Indiana.
